Researchers with Sandia National Laboratories and the U.S. Department of Energy’s (DOE) Center for Integrated Nanotechnologies in Albuquerque, New Mexico, USA, are collaborating with scientists at the Aramco Research Center in Boston, Massachusetts, USA, to better pinpoint the causes of steel pipeline corrosion in the oil and gas industry.
One of the key findings of their work1 thus far is that a localized form of nanoscale corrosion appears responsible for unpredictably decreasing the working life of steel pipes.
The researchers explain that this localized corrosion is different from uniform corrosion, which occurs in bulk form and is highly predictable. By contrast, localized corrosion is invisible and creates a pathway observable only at its end point. Based on the difficulty of detection, it becomes easier for localized corrosion to spread.
Before and After
Sandia’s expensive transmission electron microscopes could not be moved to the field. Therefore, to mimic the chemical exposure of in situ pipes, very thin pipe samples were exposed at the laboratory to a variety of chemicals known to pass through oil pipelines.
“The sample we analyzed was considered a low-carbon steel, but it has relatively high-carbon inclusions of cementite, which are the sites of localized corrosion attacks,” says Paul Kotula, a Sandia researcher. “Our microscopes were a key piece of this work, allowing us to image the sample, observe the corrosion process, and do microanalysis before and after the corrosion occurred to identify the part played by the ferrite and cementite grains and the corrosion product.”
Sandia researcher Khalid Hattar put each dry sample in a vacuum and used a microscope to create maps of the steel grain types and their orientation, with ~6 nm (0.2 µin) resolution. “By comparing these maps before and after the liquid corrosion experiments, a direct identification of the first phase that fell out of the samples could be identified, essentially identifying the weakest link in the internal microstructure,” Hattar says.
The Center for Integrated Nanotechnologies—where much of the work was performed—is a U.S. DOE Office of Science user facility operated by Sandia and Los Alamos national laboratories for university, industry, and other national lab researchers.
Microscopic Findings
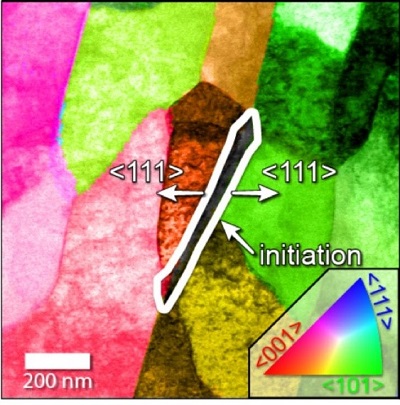 Using the microscopes, which shoot electrons through targets to take pictures, the researchers were able to pin the root of the problem on a triple junction formed by a grain of cementite, which is a compound of carbon and iron, and two grains of ferrite, a type of iron. This junction forms frequently during most methods of fashioning steel pipe.
Using the microscopes, which shoot electrons through targets to take pictures, the researchers were able to pin the root of the problem on a triple junction formed by a grain of cementite, which is a compound of carbon and iron, and two grains of ferrite, a type of iron. This junction forms frequently during most methods of fashioning steel pipe.
“This was the world’s first real-time observation of nanoscale corrosion in a real-world material—carbon steel—which is the most prevalent type of steel used in infrastructure worldwide,” says Steven Hayden, senior research scientist at Aramco. “Through it, we identified the types of interfaces and mechanisms that play a role in the initiation and progression of localized steel corrosion. The work is already being translated into models used to prevent corrosion-related catastrophes like infrastructure collapse and pipeline breaks.”
The researchers found that an interfacial disorder in the atomic structure of the triple junctions made it easier for a corrosive solution to remove iron atoms along that interface. In the experiment, the corrosive process stopped when the triple junction had been consumed by corrosion, but the crevice left behind allowed the corrosive solution to attack the interior of the steel.
“We thought of a possible solution for forming new pipe, based on changing the microstructure of the steel surface during forging, but it still needs to be tested and have a patent filed if it works,” says Sandia’s Principle Investigator Katherine Jungjohann, author of a paper on the study and lead microscopist. “But now we think we know where the major problem is.”
Overcoming Historical Limitations
When Hayden first started working in corrosion research, he “was daunted at how complex and poorly understood corrosion is.”
“This is largely because realistic experiments would involve observing complex materials like steel in liquid environments and with nanoscale resolution, and the technology to accomplish such a feat had only recently been developed and yet to be applied to corrosion,” Hayden explains. “Now, we are optimistic that further work will allow us to rethink manufacturing processes to minimize the expression of the susceptible nanostructures that render the steel vulnerable to accelerated decay mechanisms.”
The work in this study was funded in part by Sandia’s Laboratory Directed Research and Development program.
Source: Sandia National Laboratories, www.sandia.gov.
Reference
1. “Thwarting Oil-Pipeline Corrosion by Identifying a Nanoscale Villain,” Sandia Labs News Releases, June 6, 2019, https://share-ng.sandia.gov/news/resources/news_releases/thwarting-oil-pipeline-corrosion-by-identifying-a-nanoscale-villain/ (Aug. 19, 2019).
Editor’s note: Reprinted with permission from Infrastructure Insights, Summer 2019.