Although wine production is one of the oldest industries established by humankind, the modern wine industry utilizes special equipment, mostly made from austenitic stainless steel, to avoid corrosion, scale formation, and the appearance of contaminants. Additional engineering materials used are fiber-reinforced plastics, glass for bottles, and wood for tonels and casks.
In ancient cultures such as Babylonia, Egypt, Phoenicia, Israel, Greece, and Rome, vines, vineyards, and wine were part of everyday life.1 The two oldest nutritive beverages known to mankind are milk and wine, both symbolic elements in many religions.2
According to market research company International Wine and Spirits Research, global sales of wine and spirits rose to 60 billion L in 2013.3 Wine production contributes to the food industry’s position as one of the three largest markets worldwide, which also includes the energy and water industries based on production, number of consumers, and economic and social significance; and, like the energy and water industries, the wine industry is subject to corrosion.
Vine, Vineyard, and Wine
 The vine “vitis vinifera” is planted in vineyards using stem cuttings, a method brought to the Napa Valley, California, USA and the Guadalupe Valley, Baja California, Mexico by missionaries and colonists (Figures 1 and 2). Viticulture has been beneficial to the economy for these regions and many other countries. The appliances needed for the vintage are shears, trays, and baskets that must be clean to avoid diseases caused by fungi, bacteria, and insects. Human sensory examination by wine experts and tasters can detect the differences in wines (i.e., their origin, age, sugar content, flavor, and aroma). To ensure the quality of wine as well as the conservation of its organoleptic characteristics, physical and chemical analyses are applied.
The vine “vitis vinifera” is planted in vineyards using stem cuttings, a method brought to the Napa Valley, California, USA and the Guadalupe Valley, Baja California, Mexico by missionaries and colonists (Figures 1 and 2). Viticulture has been beneficial to the economy for these regions and many other countries. The appliances needed for the vintage are shears, trays, and baskets that must be clean to avoid diseases caused by fungi, bacteria, and insects. Human sensory examination by wine experts and tasters can detect the differences in wines (i.e., their origin, age, sugar content, flavor, and aroma). To ensure the quality of wine as well as the conservation of its organoleptic characteristics, physical and chemical analyses are applied.
Ethyl Alcohol
Ethyl alcohol (CH3CH2OH) is the main component of alcoholic drinks, and is produced by applying the yeast Saccharomyces to ferment sugar-containing fruits and cereal grains. Wine, the most popular drink, is obtained by anaerobic conversion of sugar in ripe grapes into ethanol, shown in Equation (1):
C6H12O6 ➞ CH3CH2OH + 2 CO2 (1)
In a subsequent stage, wine may convert into vinegar (a dilute, slightly corrosive solution of acetic acid [C2H4O2]) in another biocatalytic process, this one accomplished by the aerobic bacterium Acetobacter aceti. During this process, the previously formed ethanol is oxidized by oxygen from the air, as shown in Equation (2):
CH3CH2OH + O2 ➞ CH3COOH + H2O (2)
Therefore, wine bottles should be well-closed with impermeable, resilient, hydrophobic corks to avoid oxygen penetration that would ruin the wine. Wine bottling is done with automatic stainless steel (SS) machines (Figure 3).
Wine Production
 Modern technologies are involved in wine production. Wineries, winemakers, and wine inventories utilize software for management, production, and marketing to assure high efficiency, cost control, and profit on a global scale.4 Voltammetry and spectroscopy are applied to predict the presence of pigment and antioxidant species in the red wines of a winery in Baja California.5 The wines’ oxidation-reduction potential (ORP) is measured with a conventional potentiostat controlled by a computer. This method follows ASTM D14986 for determining the ORP of water. During safe wine transportation, microchips embedded in the crates monitor temperature and track location.
Modern technologies are involved in wine production. Wineries, winemakers, and wine inventories utilize software for management, production, and marketing to assure high efficiency, cost control, and profit on a global scale.4 Voltammetry and spectroscopy are applied to predict the presence of pigment and antioxidant species in the red wines of a winery in Baja California.5 The wines’ oxidation-reduction potential (ORP) is measured with a conventional potentiostat controlled by a computer. This method follows ASTM D14986 for determining the ORP of water. During safe wine transportation, microchips embedded in the crates monitor temperature and track location.
Wine production requires large quantities of water for cleaning, bottling, and hygienic procedures. Scaling deposited in production vessels is removed with mild acids, such as phosphoric and citric. The use of SS controls the formation of corrosion products, while strict adherence to sanitation procedures prevents contaminants. In so doing, the taste and organoleptic characteristics of the wine are conserved.
 These SS vessels are manufactured from polished sheets with smooth, reflective surfaces commonly used for food equipment.7 These finishes are achieved by chemical or electrochemical treatment, and these techniques are also applied for rehabilitation of damaged surfaces (Figure 4)
These SS vessels are manufactured from polished sheets with smooth, reflective surfaces commonly used for food equipment.7 These finishes are achieved by chemical or electrochemical treatment, and these techniques are also applied for rehabilitation of damaged surfaces (Figure 4)
In the wine industry, bisulfites are added to prevent undesirable biochemical processes. The most corrosive agent found in wine is sulfur dioxide (SO2), which can form sulfurous acid (H2SO3). SO2 provides useful properties in wine making: it is an antiseptic, it protects the wine from the deleterious effect of oxygen, and it destroys the enzyme that causes enzymatic browning.
Winery Equipment and Materials
In the past, fermentation vats, classical tonels, casks, and barrels constructed of hooped staves were made from special woods (e.g., oak), which would impart the characteristic body, taste, aroma, and color to the wine inside.
 As part of modern science and technology, an extended range of engineering materials has been developed and is presently used in the wine industry to improve the equipment and facilities. These materials encompass metals, alloys, plastics, glasses, and composites. Among their features is increased corrosion resistance and improved mechanical strength. Most of the equipment used in these plants, including hoses, pipes, pumps, tanks, fermentation vats, steamers, pasteurizers, refrigerators, blenders, filters, and packaging/bottling machines, is made of austenitic SS (e.g., UNS S31600 and S30400) to minimize corrosion. One of the world’s largest wineries is situated in Modesto, California. This winemaker is a pioneer in the use of austenitic SS tanks, which are easily cleaned, resist staining/corrosion from wine, and allow precise temperature control during fermentation.
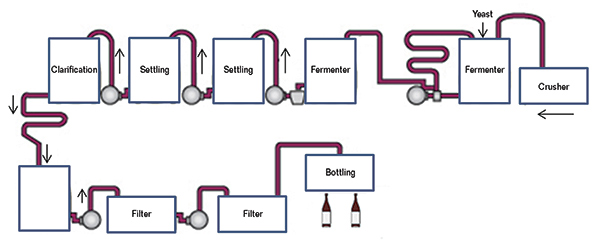
The main types of equipment installed in a modern winery are listed in Table 1, and a wine production schematic diagram is shown in Figure 5.
As part of modern science and technology, an extended range of engineering materials has been developed and is presently used in the wine industry to improve the equipment and facilities. These materials encompass metals, alloys, plastics, glasses, and composites. Among their features is increased corrosion resistance and improved mechanical strength. Most of the equipment used in these plants, including hoses, pipes, pumps, tanks, fermentation vats, steamers, pasteurizers, refrigerators, blenders, filters, and packaging/bottling machines, is made of austenitic SS (e.g., UNS S31600 and S30400) to minimize corrosion. One of the world’s largest wineries is situated in Modesto, California. This winemaker is a pioneer in the use of austenitic SS tanks, which are easily cleaned, resist staining/corrosion from wine, and allow precise temperature control during fermentation.
The main types of equipment installed in a modern winery are listed in Table 1, and a wine production schematic diagram is shown in Figure 5.
A main component of wine is ethanol, which is also used as a gasoline additive that is produced from sugar and cellulose-containing vegetables in industrial plants. Nickel-containing SS (e.g., UNS S30400, S34003, S31600, and S31603) is widely used in these plants,8 and this experience has been adapted by the wine industry.
Corrosion Prevention and Control
 Corrosion avoidance requires the application of appropriate methods and techniques from the early stages of design through the construction, erection, and operation of a full winery.9-10
Corrosion avoidance requires the application of appropriate methods and techniques from the early stages of design through the construction, erection, and operation of a full winery.9-10
Cathodic protection (CP) is employed to protect water storage tanks, cooling systems, and pipelines. Normally, CP is applied in conjunction with paints and coatings as part of an anticorrosion system.
To ensure the purity and quality of the wine, cleanliness and durability of the equipment is essential. Hygiene and sanitation are basic requirements. Equipment surfaces are restored by manual and automated mechanical or chemical cleaning, with the latter referred to as “cleaning in place” without disassembling the equipment. Sanitation and cleaning/disinfecting of the vessels and machinery in the United States proceeds in accordance with the regulations of the National Sanitation Foundation (NSF),11 which stipulates that materials in the food industry, including the wine industry, should have a smooth surface and be corrosion resistant, non-toxic, stable, and nonabsorbent.
 Mild organic acids (tartaric, malic, lactic, and citric) are present in grapes and wine, in small amounts, and alter the taste and color as well as lower the pH of the wine. Sometimes these acids crystallize and settle in the bottom of the bottle. Corrosion resistance is the main property to be considered in the choice of materials for winery equipment, but the final selection must be a compromise between technological, functional, and economic factors. Sometimes it is more economical to use a higher priced material than a lower priced material that may require frequent maintenance or replacement; however, the quality of the finished product must be paramount.
Mild organic acids (tartaric, malic, lactic, and citric) are present in grapes and wine, in small amounts, and alter the taste and color as well as lower the pH of the wine. Sometimes these acids crystallize and settle in the bottom of the bottle. Corrosion resistance is the main property to be considered in the choice of materials for winery equipment, but the final selection must be a compromise between technological, functional, and economic factors. Sometimes it is more economical to use a higher priced material than a lower priced material that may require frequent maintenance or replacement; however, the quality of the finished product must be paramount.
Conclusions
Wine consumption is being expanded due to the rise of the standard and quality of life worldwide. Wineries use tonels, casks, and barrels fabricated from classic wood materials, along with modern materials such as corrosion-resistant SS and fiber-reinforced plastics for equipment and facilities.To ensure the efficiency and profitability of the industry, the production and storage equipment should be corrosion-resistant to control the expense of maintenance and replacement of corroded equipment.
References
- M. Schorr, B. Valdez, A. Eliezer, “The Chemistry of Wine,” Medium Chemistry Magazine 98 (2015): p. 24.
- H. Riedel, et al., “Wine as Food and Medicine,” Scientific, Health and Social Aspects in the Food Industry, B. Valdez, M. Schorr, R. Zlatev, eds. (Rijeka, Croatia: InTech, 2012), pp. 399-418.
- “The Spirits Business, Cheers to Uncle Sam,” The Economist, November 29, 2014, http://www.economist.com/news/business/21635022-emerging-markets-are-grim-global-spirits-firms-america-looks-good-cheers-uncle-sam (June 16, 2015).
- “Wine Industry Software Guide” Wine Business Monthly, October 2007, http://www.winebusiness.com/wbm/?go=getArticleSignIn&dataId=51359 (June 16, 2015).
- G. Guzman, M. Schorr, B. Valdez, A. Martinez, “Voltamperometric Studies of Baja California Red Wines,” The Ocean, The Wine, and The Valley: The Lives of Antoine Badan, E.G. Pavia, J. Sheinbaum, J. Candela, eds. (Ensenada, Mexico: CICESE, 2010).
- ASTM D1498-13, “Standard Test Method for Oxidation-Reduction Potential of Water” (West Conshohocken, PA: ASTM International, 2013).
- B. Valdez, et al., “Corrosion in the Food Industry and Its Control,” Food Industrial Processes, Methods and Equipment (Rijeka, Croatia: InTech, 2012).
- “Really Green Ethanol Enabled by Nickel-Containing Alloys,” Nickel Magazine 29, 2 (2014): pp. 8-9.
- R. Raichev, L. Veleva, B. Valdez, Corrosión de Metales y Degradación de Materiales, M. Schorr, ed. (Baja California, Mexico: Universidad Autónoma de Baja California, 2009), pp. 216-233.
- B. Valdez, et al., “Corrosion Control in Industry ,” Environmental and Industrial Corrosion, B. Valdez, M. Schorr, eds. (Rijeka, Croatia: InTech, 2012): pp. 39-43.
- The National Sanitation Foundation (NFS), http://www.nfs.org ( June 16, 2015).
B. VALDEZ is a full professor at the Institute of Engineering, Universidad Autónoma de Baja California, Blvd. Benito Juárez y Calle de la Normal s/n, Col. Insurgentes Este, C.P. 21280, Mexicali, BC, Mexico, e-mail: benval@uabc.edu.mx. He has a B.Sc. in chemical engineering, a M.Sc. and a Ph.D. in chemistry, and is a member of the Mexican Academy of Science and the National System of Researchers in Mexico. He was the guest editor of Corrosion Reviews, in which he produced two special issues on corrosion control in geothermal plants and the electronics industry. His activities include corrosion research, consultancy, and control in industrial plants. During the IMRS Congress in August 2013 in Mexico, he received a National Distinguished Career Award from NACE International and the NACE Central Mexico Section. He has been a NACE International member for 26 years.
M. SCHORR is a professor (Dr. Honoris causa) at the Institute of Engineering, Universidad Autónoma de Baja California, e-mail: mschorr2000@yahoo.com. He has a B.Sc. in chemistry, a M.Sc. in materials engineering from the Technion-Israel Institute of Technology, and 50 years of experience in environmental and industrial corrosion control. From 1986 to 2004, he was the editor of Corrosion Reviews. He has published 354 scientific and technical articles in English, Spanish, and Hebrew on materials and corrosion. He has worked as a corrosion consultant and professor in Israel, the United States, Latin America, Spain, and South Africa. During The IMRS Congress in August 2010, in Mexico, he received an International Distinguished Career Award from NACE International and the NACE Central Mexico Section. He is a member of the National System of Researchers in Mexico. He has been a NACE International member for 23 years.
N. LOTAN has been a full professor in the Department of Biomedical Engineering since 1989 at the Technion-Israel Institute of Technology, Haifa, Israel. He joined the Technion-IIT in 1979. He held visiting appointments at the National Center for Scientific Research in Strasbourg, France; Cornell University in Ithaca, New York, USA; the University of Rouen in France; the Ciba-Geigy Corp. in Basel, Switzerland; and the Massachusetts Institute of Technology in Boston, Massachusetts, USA. During 2006, Lothan taught a course on advanced materials at the University of Baja California, Mexico. Lotan is the incumbent of The Roy Matas/Winnipeg Chair in Biomedical Engineering, and is the laboratory coordinator of the Leonard and Diane Sherman Biomaterials Research Center.
A. ELIEZER is the director of the Corrosion Research Center, Nano-Bio & Advanced Materials, as well as a faculty member of both civil engineering and mechanical engineering at the Sami Shamoon College of Engineering, Ber Sheva, Israel. He is the director of the Research and Development Authority. He is active in the NACE Europe Area, is a board member of the World Corrosion Organization, is an EFC member and CAMPI chair, as well as a faculty advisor of the NACE Israel Negev Student Section.